Medical implant devices are a high-risk product group because their materials remain in long-term contact with living tissue, bodily fluids, and blood. Therefore, rubber used for implants must meet stringent requirements for biocompatibility, long-term stability, and control of extractables and leachables. In this article, Thai Duong Plastics compiles key technical requirements and important standards to help select suitable rubber materials for medical implant devices.
Why does rubber for implant devices require its own standards?
Rubber used in implant devices requires its own standards because the device is placed inside the human body for an extended period, rather than only having short-term contact like many common medical devices. Throughout use, the material must remain in continuous contact with living tissue and biological fluids under the body’s stable temperature and humidity conditions.
Unlike disposable gloves or single-use tubing, implant devices require materials that are not only safe at the time of use but also maintain long-term stability. If the rubber ages, loses elasticity, or releases undesired substances over time, the device may trigger inflammation or fail to maintain its original function.
For that reason, rubber for implant devices must be evaluated and controlled under stricter standards, focusing on long-term biological evaluation, control of extractables and leachables, and the ability to maintain safety and performance throughout the product lifecycle.

Distinguishing general medical rubber from implant-grade rubber
In practice, “medical grade” is often used to describe materials suitable for medical applications in general. This group of rubbers is primarily designed for devices with short- or medium-term contact with the human body, such as consumables or components that do not remain inside the body, and typically only need to meet baseline biocompatibility requirements.
In contrast, rubber used for implant devices is commonly considered “implant grade,” meaning it is selected for long-term contact with living tissue and biological fluids. This material class requires deeper biological and chemical data, including control of extractables and leachables and the ability to maintain stability throughout the service life. Therefore, you cannot rely solely on a “medical grade” description or USP Class VI results to conclude suitability.
Technical requirements for rubber materials used in implant devices
To ensure safety and effectiveness in implant applications, rubber materials must meet multiple technical requirements throughout the device lifecycle:
Biological evaluation requirements
Biological evaluation is mandatory when using rubber for implant devices because the material has direct and long-term contact with the human body. The evaluation is not only to confirm that the material is safe at the time of use, but also to assess compatibility over the entire period the device exists in a biological environment.
Under the standard approach, biological evaluation is built based on the type and duration of body contact. For implant devices in the long-term contact category, rubber materials often need to be assessed for cytotoxicity, irritation, sensitization, systemic toxicity, and responses at the implantation site. The test scope must be defined based on risk management and international biocompatibility standards (such as ISO 10993).
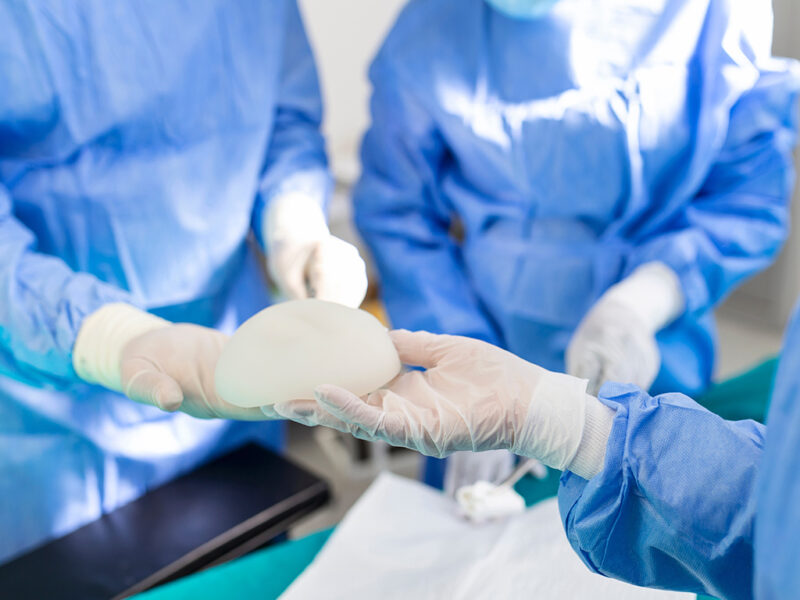
Chemical control requirements
In addition to biological evaluation, chemical control is a key factor for rubber materials used in implant devices, especially in long-term contact applications. During manufacturing and use, rubber may release unwanted substances if not properly controlled. Therefore, chemical control requirements typically focus on:
- Assessing extractables and leachables (E&L) to control hazardous impurities that may be released into the body.
- Establishing safe exposure limits based on toxicological assessment and supporting analytical data.
- Linking chemical data to the risk file to manage overall device safety.
Accordingly, in medical device engineering, materials are not evaluated based on a subjective “non-toxic” claim; they must be proven to have controlled extractables and leachables within safe limits under applicable standards.
Requirements for long-term stability and mechanical/physical performance
Implant devices must maintain stable function throughout the time they remain in the body. Therefore, the rubber used must ensure long-term mechanical/physical stability in a biological environment. Commonly considered factors include tensile strength, elongation, elasticity, compression set (permanent deformation), fatigue resistance, and crack resistance-so the device can retain stable performance under continuous physiological loading without mechanical deformation or failure.
Compatibility with sterilization processes
Sterilization compatibility does not only mean the material can withstand steam sterilization, EtO gas, radiation, etc. It must also be demonstrated that the material’s mechanical/physical, biological, and chemical properties do not change to a degree that creates risk after sterilization. In many cases, evaluation needs to combine sterilization and aging to reflect real-world use conditions.

Rubber families commonly considered for implant devices
Not every medical-grade rubber is suitable for long-term implant applications. Silicone rubber is a leading option thanks to its inertness and high biocompatibility. In particular, liquid silicone rubber (LSR) is increasingly preferred for implant components due to its high purity and excellent precision in processing.
However, even for silicone, implant use must still be based on a specific biological and chemical evaluation dossier for each application. Other elastomers may be suitable for certain medical purposes, but when considering long-term contact, data requirements and risk controls typically narrow the range of viable options.
Technical documentation requirements for rubber materials used in implants
To reduce risk during device development, reviewing the material dossier is a non-negotiable step. Technical documentation for rubber used in implant devices typically should include:
- A clear declaration of compliance and defined scope of application.
- Biological evaluation data (ISO 10993) appropriate for long-term contact.
- Chemical test reports for extractables and leachables.
- Compatibility data for the intended sterilization method.
- Information on formulation change control.

Rubber materials for medical implant devices must be selected using a comprehensive approach that integrates biological evaluation, chemical control, long-term mechanical/physical stability assessment, and risk management. Properly understanding the scope of each standard and controlling materials from the design stage helps minimize risks and improves the safety and reliability of the device throughout its service life.
