
Plastics play an important role and are among the most widely used materials in most manufacturing and everyday applications. Not only because they are affordable, plastics also offer outstanding characteristics such as being lightweight, durable, flexible, and easy to shape. To better understand this material, join Thai Duong Plastics as we explore the article below.
What Are Plastics and Where Do They Come From?
Plastic is an organic material made primarily of polymer molecules. A polymer is a long chain of many smaller units called monomers. When monomers bond together through chemical reactions (mainly polymerization or polycondensation), they form large molecular chains that are stable and flexible.
The term “plastic” originates from the Greek word “plastikos,” meaning “capable of being molded.” This accurately reflects the nature of plastics: they can be easily molded, pressed, blown, drawn, and formed into many different shapes depending on usage needs.

The Formation and Development of Plastics
Natural plastics have been used by humans since very early times-typical examples include rubber and pine resin. However, synthetic plastics truly emerged in the 19th century, when scientists discovered how to synthesize cellulose nitrate.
A new era for materials science began in 1907 with the invention of Bakelite, the first fully synthetic plastic and a thermosetting material. In the 20th century, a series of modern plastics such as PE, PP, PVC, and more were developed, making plastics indispensable in everyday life. Petroleum and natural gas quickly became the main raw materials used to mass-produce various plastics with different properties.
Today, along with the development of green technology, the use of bioplastics and recycled plastics is being promoted to reduce dependence on petroleum and limit negative environmental impacts.
What Is Plastic Made From?
Plastics are produced mainly from hydrocarbon-based raw materials such as petroleum and natural gas, with a smaller portion derived from coal or biomass (cellulose).
During processing, crude oil is separated into lighter molecules called monomers, such as ethylene, propylene, and styrene. These are the building blocks used to synthesize different plastics through polymerization reactions.
- Example of another raw-material source: Salt (NaCl) can be electrolyzed to produce chlorine, a key component for making vinyl chloride monomer.
In addition to polymers as the main component, plastics are also blended with various additives to improve mechanical performance, color durability, and heat resistance. Common additives include plasticizers, mineral fillers, colorants, and flame retardants.
The Chemical Formula of Plastics
There is no single fixed chemical formula for all plastics, because each type has its own molecular structure. However, most plastics are formed from polymer chains primarily composed of carbon and hydrogen, sometimes with additional elements such as oxygen, chlorine, sulfur, and nitrogen.
A general formula often used to represent many common vinyl plastics (polymers formed through addition polymerization) is (CH2-CHX)n, where X is an atom or functional group specific to each plastic type. Some specific examples include:
| Plastic Type | Monomer | Polymer Formula | Common Applications |
| Polyethylene (PE) | Ethylene (CH2=CH2) | (CH2-CH2)n | Plastic bags, wrapping film, bottles and containers. |
| Polypropylene (PP) | Propylene (CH2=CHCH3) | (CH2-CHCH3)n | Packaging, food containers, engineering parts. |
| Polyvinyl Chloride (PVC) | Vinyl chloride (CH2=CHCl) | (CH2-CHCl)n | Water pipes, electrical cables, construction materials. |
| Polystyrene (PS) | Styrene (CH2=CHC6H5) | (CH2-CHC6H5)n | Plastic cups, foam boxes, household goods. |
| Polytetrafluoroethylene (PTFE) (Teflon) | Tetrafluoroethylene (CF2=CF2) | (CF2-CF2)n | Non-stick coatings for pans, electronic components. |
These polymer chains can be arranged in different structures, creating a wide range of strength, elasticity, heat resistance, and transparency across plastic types. Differences in formulas and bonding between monomers are what allow plastics to exist in many forms and serve many different purposes.
Structure and Properties of Plastics
Plastics consist of two main parts: the base polymer (which determines core properties) and additives (which help adjust performance). Plastics have low density, making them very lightweight, and they offer electrical insulation, thermal insulation, and strong corrosion resistance. Plastics do not rust and can withstand the effects of many chemicals.
Depending on polymer structure, plastics are divided into two main categories:
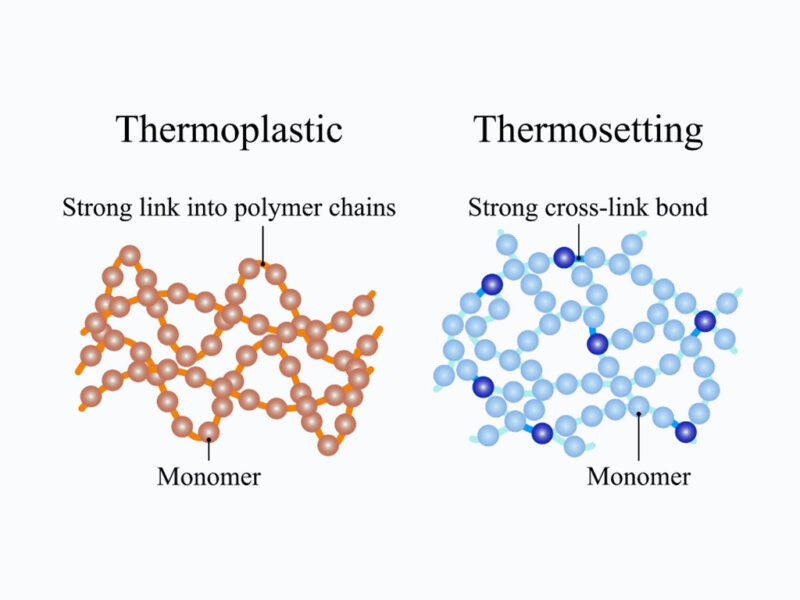
- Thermoplastics (Thermoplastics):
Common examples include PE, PP, PET, and PS. When heated, they become soft and pliable, can be shaped in molds, and harden again upon cooling. They can be recycled by melting and remolding.
- Thermosets (Thermosets):
Types such as Bakelite, Epoxy, and Melamine-after being shaped and cured using heat or catalysts-form permanent, durable cross-links. They cannot soften again when reheated and are difficult or impossible to recycle using conventional melting methods.
Advantages and Limitations of Plastics
Thanks to their flexibility and high adaptability, plastics have become indispensable in modern life. However, alongside their outstanding advantages, plastics also come with notable limitations that should be clearly understood to ensure appropriate use and handling.
Advantages of plastics:
- Lightweight, easy to transport, and cost-effective to manufacture.
- High mechanical strength, impact resistance, and good corrosion resistance.
- Effective electrical insulation, thermal insulation, and water resistance.
- Easy to shape, enabling production in various sizes, designs, and colors.
- Plastic processing is relatively simple and consumes less energy than metals or glass.
- Can be combined with additives to modify properties based on usage requirements.
Limitations of plastics:
- Slow to decompose in nature, causing environmental pollution if not handled properly.
- Production depends heavily on petroleum and natural gas, which are non-renewable resources.
- Some plastics may release toxic substances when exposed to high temperatures.
- Recycling remains challenging due to differences in properties across plastic types.

Plastics are materials formed from organic compounds derived primarily from petroleum and natural gas, then polymerized to create durable, flexible polymer chains. Thanks to the ability to incorporate additives, plastics can be adapted to a wide range of applications. However, along with these significant benefits, plastics also present major environmental challenges. Proper use, increased recycling, and the development of bioplastics are necessary pathways to balance economic value with ecosystem protection.
See more: Top 11 Plastic Injection Molding Companies in Vietnam
